2024
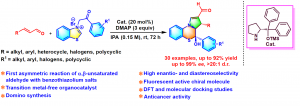
141. Organocatalyzed Enantio- and Diastereoselective Formal Domino 1,3-Dipolar Cycloaddition/Rearrangement: Synthesis of Chiral Pyrrolo-thiazine-2-carbaldehydes (Solai Pandidurai, Venkata Surya Kumar Choutipalli, Venkatesan Subramanian and G. Sekar, Org. Lett., 2024, 26, 15, 2971-2975. (https://doi.org/10.1021/acs.orglett.4c00544)
2023
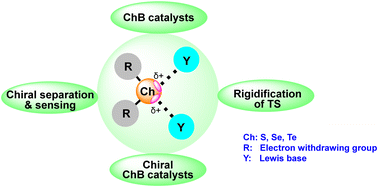
140. Chemical Society Reviews: Chalcogen bonding catalysis (G. Sekar, Vysakh V Nair, Jieping Zhu, Chem. Soc. Rev,.2024, 53, 586). https://doi.org/10.1039/D3CS00503H
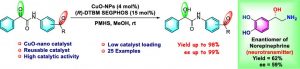
139. Chemo- and Enantioselective Reduction of α-Keto Amides to α-Hydroxy Amides using Reusable CuO-Nanoparticles as Catalyst (Gollapalli Narasimha Rao, G. Sekar, J. Org. Chem.,2023, 88, 4008- 4016).https://doi.org/10.1021/acs.joc.3c00090
138. Binaphthyl-Stabilized Palladium Nanoparticle-Catalyzed Stereoselective Synthesis of 3-Arylidene-2-oxindoles via One-Pot Heck-like Carbocyclization/Nucleophilic Addition (Naziya Parveen, Solai Pandidurai, and G. Sekar, J. Org. Chem., 2023, 88, 3, 1730-1741. (https://doi.org/10.1021/acs.joc.2c02792)
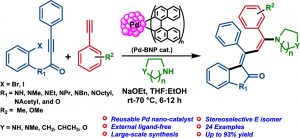
137. Stable and reusable Pd-nanoparticle catalyzed synthesis of symmetrical and unsymmetrical 1,2-dicarbonyl compounds (Gollapalli Narasimha Rao, G. Sekar, New J. Chem., 2023, 47, 3167-3177, One of the most read articles in Mar- Apr.2023). (https://doi.org/10.1039/D2NJ05538D)
2022
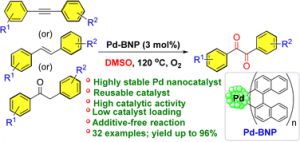
136. Halogen Bond-Activated Visible-Light-Mediated Regioselective C–H Arylation of 2-Phenylimidazo-[1,2-a]pyridines (Imran Kazi, Anuradha Nandy, Raji Selvam, G. Sekar, J. Org. Chem. 2022, 87, 18, 12323–12333, https://doi.org/10.1021/acs.joc.2c01548
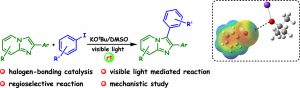
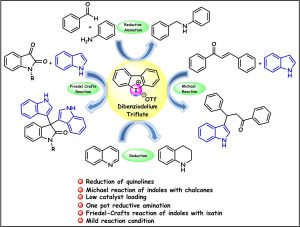
135. Dibenziodolium Salts as Halogen Bond Donor Catalysts for Reduction of Quinolines, One-Pot Reductive Amination, and Addition Reaction with Indoles (Anuradha Nandy and G. Sekar, Eur. J. Org. Chem. 2022, (https://doi.org/10.1002/ejoc.202200982)
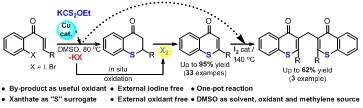
134. Cu-Catalyzed and Iodine Mediated Synthesis of Thioaurones via In-situ C-S Bond Generation using Xanthate as Sulfur Surrogate (P. Soundarya and G.Sekar, Biomol. Chem. 2022, 20, 7405 (DOI: 10.1039/d2ob01211a)
133. Synergistic Dual Amine/Transition Metal Catalysis-Recent Advances, (V. V. Nair, D. Arunprasath, S. Pandidurai, G. Sekar, Eur. J. Org. Chem. 2022, In Press (https://doi.org/10.1002/ejoc.202200244) (Invited Review)

132. Transition metal-free iodine-catalyzed denitrative C–S cross-coupling: An atypical route to access thiochromane derivatives (Anuradha Nandi and Sekar, J. Org. Chem. 2022, DIO: 10.1021/acs.joc.2c00425).

131. Iodine-promoted controlled and selective oxidation of (aryl)(heteroaryl)methanes (Somraj Guha, Imran Kazi, D. Sathish, and G. Sekar, J. Org. Chem. 2022, 87, 5424)

2021
130. KOtBu Promoted Halogen Bond Assisted Intramolecular C-S Cross-Coupling of o-iodothioanilides for the Synthesis of 2-Substituted Benzothiazoles (Anuradha Nandi and G. Sekar, J. Org. Chem. 2021, 86, 15825) (Link)

129. Copper-Catalyzed Domino Synthesis of Multisubstituted Benzo[b]thiophene Through Radical Cyclization Using Xanthate as Sulfur Surrogate (N. Sundaravelu, Tushar Singha, Anuradha Nandy, and G. Sekar, Chem. Commun. 2021, 57, 4512) (Link)
128. Visible Light Mediated Photocatalyst Free C-S Cross-Coupling: Domino Synthesis of Thiochromane Derivatives via Photoinduced Electron Transfer (N. Sundaravelu, Anuradha Nandy, and G. Sekar, Org. Lett., 2021, 23, 3115–3119) (Link)

127. Visible Light-Driven Halogen Bond-Assisted direct synthesis of heteroaryl thioethers using Transition Metal-Free one-pot C-I bond formation/ C-S cross-coupling reaction (A. Nandy, I. Kazi, S. Guha, and G. Sekar, J. Org. Chem. 2021, 86, 2570) (Link)

126. Metal-catalyzed C-S bond formation using sulfur surrogates (N. Sundaravelu, S. Sangeetha, and G. Sekar, Org. Biomol. Chem., 2021, 19, 1459) (Review) (Link)
2020
125. Copper-catalyzed double C-S bond formation for the synthesis of 2-acyldihydrobenzo[b]thiophenes and 2-acylbenzo[b]thiophenes (S. Sangeetha and G. Sekar, Chem. Commun., 2020, 56, 10906).
124. Palladium Nanoparticles-Catalyzed Stereoselective Domino Synthesis of all Carbon Tetrasubstituted Olefin Containing Oxindoles via Carbopalladation/C-H Activation (Naziya Parveen and G. Sekar, J. Org. Chem. 2020, 16, 10514).

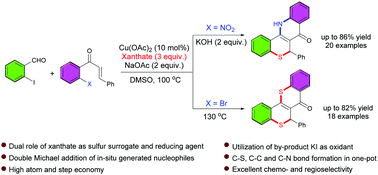
123. Cu-Catalyzed one-pot synthesis of thiochromenoquinolinone and thiochromeno-thioflavone via oxidative double hetero Michael addition using in situ generated nucleophiles† (N. Sundaravelu and G. Sekar, Chem. Commun., 2020, 56, 8826).
122. Iodonium Ion-Catalyzed Domino Synthesis of Z‑Selective α,β-Diphenylthio Enones from Easily Accessible Secondary Alcohols (N. Sundaravelu, Somraj Guha, and Sekar, J. Org. Chem. 2020, 85, 5895-5906).

121. Palladium Nanoparticles-Catalyzed Stereoselective Domino Synthesis of 3-Allylidene-2(3H)-oxindoles and 3-Allylidene-2(3H)-benzofuranones (Naziya Parveen and Sekar, J. Org. Chem. 2020, 85, 4682-4694 ).

2019
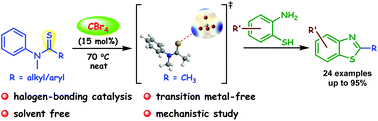
120. An efficient synthesis of benzothiazole using tetrabromomethane as a halogen bond donor catalyst (Imran Kazi and G. Sekar, Org. Biomol. Chem., 2019, 17, 9743).
119. Surface enriched palladium on palladium-copper bimetallic nanoparticles as catalyst for polycyclic triazoles synthesis (Rajib Saha and Sekar, J. Catal. 2019, 377, 673).

118. Domino Synthesis of Thioflavones and Thioflavothiones by Regioselective Ring Opening of DonorAcceptor Cyclopropane Using In-Situ-Generated Thiolate Anions (N. Sundaravelu, and Sekar, Org. Lett., 2019, 21, 6648).

117. Palladium Nanoparticles-Catalyzed Synthesis of Indanone Derivatives via Intramolecular Reductive Heck Reaction (Naziya Parveenaand and Sekar, Adv. Synth. Catal., 2019, 361, 4581).

116. Ligand-Free and Reusable Palladium Nanoparticles-Catalyzed Alkylation of 2-Alkylazaarenes with Activated Ketones under Neutral Conditions (Naziya Parveen, A. Muthukumar, and Sekar, Adv. Synth. Catal., 2019, 361, 4255).

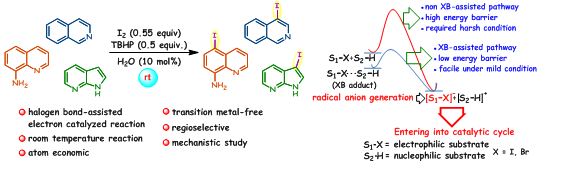
115. Halogen Bond–Assisted Electron–Catalyzed Atom Economic Iodination of Heteroarenes at Room Temperature (I. Kazi, S. Guha, and G. Sekar, J. Org. Chem. 2019, 84, 6648 (One of the most read articles in May-June 2019) (Link)
113. A covalently linked dimer of [Ag25(DMBT)18]-( Md Bodiuzzaman, A Nag, R. N.Pradeep, A. Chakraborty, R. Bag, P. Ganesan, G. Natarajan, G. Sekar, S. Ghosh and T. Pradeep, Chem. Commun., 2019, 55, 5025. (Link)
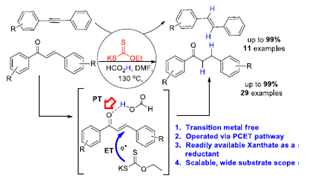
112. Proton-Coupled Electron Transfer: Transition-Metal-Free Selective Reduction of Chalcones and Alkynes Using Xanthate/Formic Acid (R.Prasanna, Somraj Guha, and G. Sekar, Org. Lett., 2019, 21, 2650.) (Link)
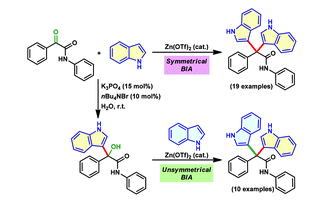
111. Zn(OTf )2-catalyzed access to symmetrical and unsymmetrical bisindoles from αketo amides (A.Muthukumar, G. Narasimha Rao and G. Sekar , Org. Biomol. Chem., 2019, 17, 3921). (Link)
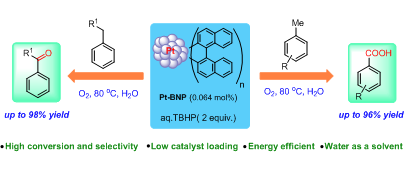
110. Selective Oxidation of Alkylarenes to Aromatic Acids/Ketone in Water by Using Reusable Binaphthyl Stabilized Pt Nanoparticles (Pt-BNP) as Catalyst (Rajib Saha and G. Sekar, Applied Catalysis B: Environmental, 2019, 250, 325.) (Link) (doi.org/10.1016/j.apcatb.2019.03.052).
(This research work is highlighted in “The Hindu” national English newspaper on 31st March
2019 (Page 13 of Chennai Edition)
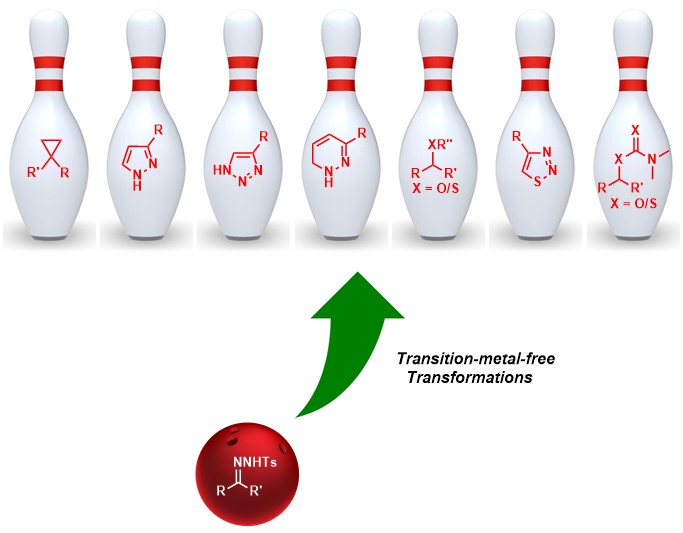
109. Luxury of N-Tosylhydrazones in Transition-Metal-Free Transformations (D. Arunprasath, B. Devi Bala and G. Sekar, Adv. Synth. Catal., 2019, 361, 1172 ).(Review Article). (Link)
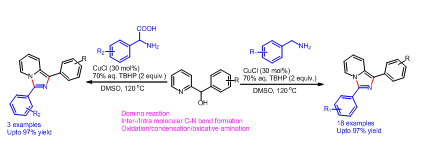
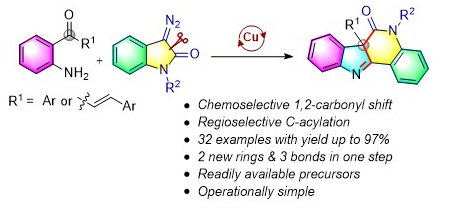
108. Copper(II)-Catalyzed Domino Synthesis of Indolo[3,2- c]quinolinones via Selective Carbonyl Migration (D. Arunprasath, and G. Sekar, Org. Lett., 2019, 21, 867.) (Link)
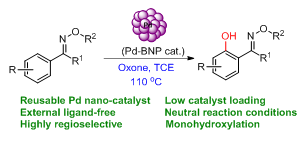
107. Reusable Palladium Nanoparticles Catalyzed Oxime Ether Directed Mono Ortho-Hydroxylation under Phosphine Free Neutral Condition (R. Saha, N. Parveen, N. Nihesh, and G. Sekar, Adv. Synth. Catal., 2019, 361, 510.) (Link)
106. Copper-Catalyzed One-Pot Synthesis of 2-Arylthiochromenones: An in Situ Recycle of Waste Byproduct as Useful Reagent (S. Sangeetha, and G. Sekar, Org. Lett., 2019, 21, 75). (Link)
2018
105. Dual Role of N-Bromosuccinimide as Oxidant and Succinimide Surrogate in Domino One-Pot Oxidative Amination of Benzyl Alcohols for the Synthesis of α–Imido Ketones (M. Muneeswara, A. Muthukumar, G. Sekar, ChemistrySelect, 2018, 3, 12524). (Link)
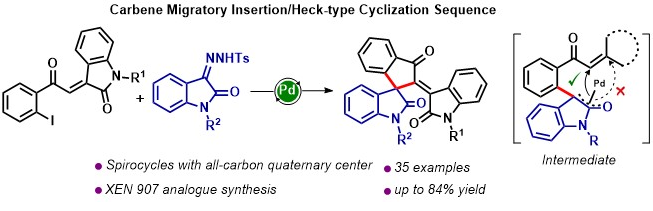
103. Stable Pd-Nanoparticles Catalyzed Domino C-H Activation/C-N Bond Formation Strategy: An Access to Phenanthridinones (Rajib Saha and G. Sekar, J. Catalysis, 2018, 366, 176-188). (Link)
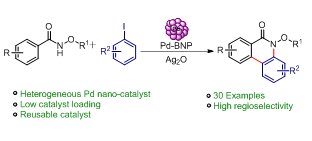
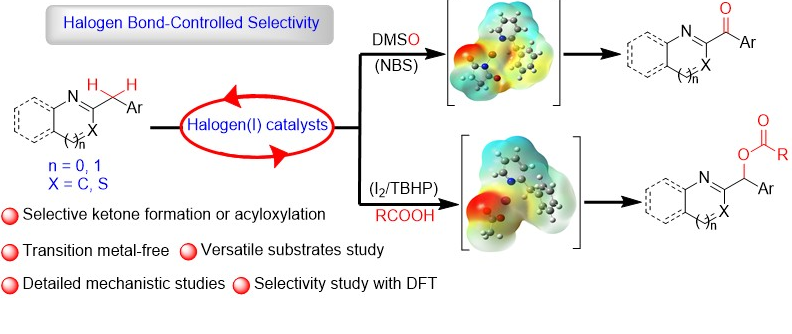
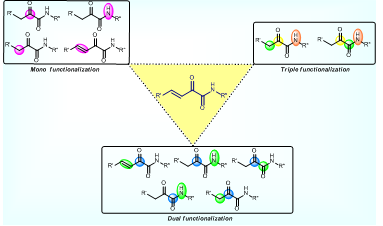
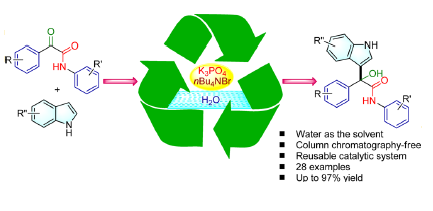
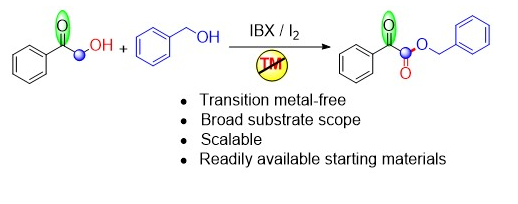
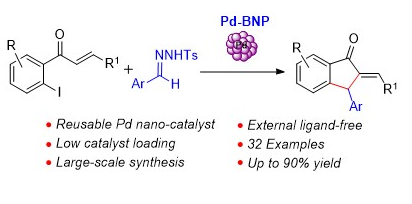
96. Stereoselective Construction of α-Tetralone-Fused Spirooxindoles via Pd-Catalyzed Domino Carbene Migratory Insertion/Conjugate Addition Sequence (D. Arunprasath, B. Devi Bala and G. Sekar, Org. Lett., 2017, 19, 5280 (One of the most read articles in Sep-Oct ’17; Highlighted in Synfacts) (Link)
95. Halogen–bonded Iodonium Ion Catalysis: A Route to α–Hydroxy Ketone via Domino Oxidations of Secondary Alcohol and Aliphatic C–H Bond with High Selectivity and Control (Somraj Guha, Imran Kazi, Pranamita Mukherjee and G. Sekar, Chem. Commun., 2017, 53, 10942) (Link)
94. Reusable Palladium Nanoparticles Catalyzed Conjugate Addition of Aryl Iodides to Enones: Route to Reductive Heck Product (Naziya Parveen, Rajib Saha and G. Sekar, Adv. Synth. Catal., 2017, 359, 3741) (Link)
93. Role of Lewis Base–Coordinated Halogen(I) Intermediates In Organic Synthesis: The Journey From an Unstable Intermediate to a Versatile Reagent (Invited review article) (Somraj Guha, Imran Kazi, Anuradha Nandy and G. Sekar, Eur. J. Org. Chem., 2017, 5497 (One of the most accessed papers in the month of 10/2017; Highlighted in Outstanding Organics) (Link)
92. NIS Mediated Cross-coupling of C(sp2)-H and N-H Bonds: A Transition Metal-free approach towards Indolo[1,2-a]quinazolinones (B. Sindhura and G. Sekar, J. Org. Chem., 2017, 82, 7657 (One of the most read articles in June-July17) (Link)
91. A Mild and Chemoselective Hydrosilylation of α-Keto Amides using Cs2CO3/PMHS/2-MeTHF System (G. Kumar, A. Muthukumar and G. Sekar, Eur. J. Org. Chem., 2017, 4883) (Link)
90. Synthesis of 2-Acylbenzo[b]thiophenes via Cu-Catalyzed α-C-H Functionalization of 2-Halochalcones Using Xanthate (S. Sangeetha and G. Sekar, Org. Lett., 2017, 19, 1670) (Link)
89. Palladium-Nanoparticles Catalyzed Oxidative Annulation of Benzamides with Alkynes for the Synthesis of Isoquinolones (Nidhi Sharma, Rajib Saha, Naziya Parveen and G. Sekar Adv. Synth. Catal. 2017, 359, 1947). (Link)
88. CBr4 as a Halogen Bond Donor Catalyst for the Selective Activation of Benzaldehydes to Synthesize α,β-Unsaturated Ketones (Imran Kazi, Somraj Guha and G. Sekar, Org. Lett., 2017, 19, 1244).(One of the most accessed top 10 papers in Org. Lett. for the month Feb-Mar,2017) (Link)
87. Domino Synthesis of Thiochromenes through Cu-Catalyzed Incorporation of Sulfur using Xanthate Surrogate (P. Muthupandi, N. Sundaravelu and G. Sekar, J. Org. Chem., 2017, 82, 1936). (Link)
86. Zinc-Catalyzed Chemoselective Alkylation of α-Keto Amides with 2-Alkylazaarenes (A. L. Muthukumar and G. Sekar, Org. Biomol. Chem., 2017, 15, 691). (Link)
85. A Transition-Metal-Free and Base-Mediated Carbene Insertion into S-S and Se-Se Bonds: An Easy Access to Thio- and Selenoacetals (D. Arunprasath and G. Sekar, Adv. Synth. Catal., 2017, 359, 698). Highlighted in Chemistryviews (Link)
2016
84. (Review Bimetallic Chiral Nanoparticles as Catalysts for Asymmetric Synthesis article) (G. Savitha, Rajib Saha and G. Sekar, Tetrahedron Lett., 2016, 57, 5168). (Link)
83. Sulfoximinocarbonylation of aryl halides using heterogeneous Pd/C catalyst (Balasubramanian Devi Bala, Nidhi Sharma and G. Sekar, RSC Advances, 2016, 6, 97152). (Link)
82. Palladium Nanoparticles Catalyzed Aroylation of NH-Sulfoximines with Aryl Iodides (Nidhi Sharma and G. Sekar RSC Advances, 2016, 6, 37226). (Link)
81. An efficient, stable and reusable palladium nanocatalyst: Chemoselective reduction of aldehydes with molecular hydrogen in water (Surya Srinivas K, Nidhi Sharma and G. Sekar Adv. Synth. Catal., 2016, 358, 1694) (Link)
80. Iron-catalyzed one-pot N-aroylation of NH‑sulfoximines with methyl arenes through benzylic C-H bond oxidation (M. Muneeswara, Surya Srinivas. K and G. Sekar, Synthesis, 2016, 48, 1541). (Link)
79. An Efficient Synthesis of Iminoquinones by Chemoselective Domino ortho-Hydroxylation/oxidation/imidation Sequence of 2-Aminoaryl Ketones (S. Chandrasekar and G. Sekar, Org. Biomol. Chem., 2016, 14, 3053). (Link)
78. Stable and Reusable Platinum Nanocatalyst: An Efficient Chemoselective Reduction of Nitroarenes in Water (Surya Srinivas K., Nidhi Sharma and G. Sekar, Tetrahedron Lett.,2016, 57, 1410). (Link)
77. Iodine mediated intramolecular C2-amidative cyclization of indoles: A facile access to indole fused tetracycles (Sindhura B., V. Rajeshkumar and G. Sekar, Org. Biomol. Chem., 2016, 14, 2297). (Link)
76. Potassium phosphate catalyzed chemoselective reduction of α-ketoamides: Route to synthesize Passerini adducts and 3-phenyloxindoles (Alagesan Muthukumar, N. Chary Mamillapalli and G. Sekar, Adv. Synth. Catal. 2016, 358, 64). (Link)
75. Stable and Reusable Binaphthyl-Supported Palladium Catalyst for Aminocarbonylation of Aryl Iodides (Nidhi Sharma and G. Sekar, Adv. Synth. Catal. 2016, 358, 314). (Link)
2015
74. Cu-Catalyzed Domino Synthesis of 2-Aryl-thiochromanones through Concomitant C-S Bond Formations Using Xanthate as Sulfur Source (S. Sangeetha, P. Muthupandi and G. Sekar, Org lett, 2015, 17, 6006). (Link)
73. Enantioselective Synthesis of α-Hydroxy Amides and β-Amino Alcohols from α-Keto Amides (N Chary Mamillapalli, and G. Sekar, Chem. Eur. J., 2015, 21, 18584). (Link)
72. Palladium-Catalyzed Intermolecular Carbene Insertion Prior to Intramolecular Heck Cyclization: Synthesis of 2 Arylidene-3-aryl-1-indanones (D. Arunprasath, P. Muthupandi, and G. Sekar, Org Lett., 2015, 17, 5448). (Link)
71. Metal Free Synthesis of a-Keto Amides from 2-Oxo Alcohols through Domino Alcohol Oxidation-Oxidative Amidation Reaction (S. S. Kotha and G. Sekar, Tetrahedron Lett., 2015, 56, 6323).
70. hemoselective Reductive Deoxygenation and Reduction of α-Keto Amides by using Palladium Catalyst (N. C. Mamillapalli and G. Sekar, Adv. Synth. Catal. 2015, 357, 3273). (Link)
69. An efficient and metal free synthesis of benzylpyridines using HI through the deoxygenation reaction (S.Chandrasekar, I. Karthikeyan and G. Sekar, RSC Advances, 2015, 5, 58790). (Link)
68. Metal Free One-Pot Synthesis of α-Ketoamides from Terminal Alkenes, (Sayan Dutta, S. S. Kotha and G. Sekar, RSC Advances, 2015, 5, 47265). (Link)
67. Pd-catalyzed direct C2-acylation and C2,C7-diacylation of indoles: pyrimidine as easily removable C-H directing group (G. Kumar, G. Sekar, RSC Advances, 2015, 5, 28292). (Link)
66. Domino Reactions: Concept for efficient Organic Synthesis 2nd Editionv(L.F. Tietze, Ed.) Book chapter Title: “Oxidation and Reduction Reactions in Domino Processes (G. Sekar, I. Karthikeyan, D. Ganapathy, Wiley-VCH, Weinheim. 2014, ISBN 978-3-527-33432-2).
65. Iron-Catalyzed Direct Synthesis of Amides from methylarenes (Surya Srinivas K., Sindhura B. and G. Sekar, Adv. Synth. Catal. 2015, 357, 1437), (Link)
64. A Versatile and One-Pot Strategy to Synthesize α-Amino Ketones from Benzylic Secondary Alcohols using N-Bromosuccinimide (Somraj Guha, V. Rajeshkumar, S. Kotha Surya and G. Sekar, Org. Lett, 2015, 17, 406), (Link)
63. An Efficient Synthesis of Pyrido[1,2-a]indoles through Aza-Nazarov Type Cyclization (I. Karthikeyan, D. Arunprasath, and G. Sekar, Chem. Commun., 2015, 51, 1701). (Link)
62. Copper-Catalyzed One-Pot Synthesis of α-Ketoamides from 1-Arylethanols
(Nidhi Sharma, Nabajit Lahiri, K. Surya Srinivas and G. Sekar, Synthesis, 2015, 47, 726)
61. Stable Palladium Nanoparticles Catalyzed Synthesis of Benzonitriles Using K4[Fe(CN)6] (D. Ganapathy, Surya S. Kotha and G. Sekar, Tet. Lett. 2015, 56, 175)
2014
60. An Efficient Synthesis of Polysubstituted Olefins Using Stable Palladium Nanocatalyst: Applications in Synthesis of Tamoxifen and iso-Combretastatin A4 (D. Ganapathy and G. Sekar, Org. Lett. 2014, 16, 3856) (Link)
59. Metal free chemoselective reduction of α-keto amides using TBAF catalyst (N. C. Mamillapalli and G. Sekar, RSC Advances, 2014, 4, 61077) (Link)
58. Iron Catalyzed C−H Bond Functionalization for the Exclusive Synthesis of Pyrido[1,2-a]indoles or Triarylmethanols (I. Karthikeyan and G. Sekar, Eur. J. Org. Chem. 2014, 8055). (Link)
57. An efficient route to synthesize isatins by metal-free, iodine-catalyzed sequential C(sp3)–H oxidation and intramolecular C−N bond formation of 2′-aminoacetophenones (Rajesh Kumar, S. Chandrasekar and G. Sekar, Org. Biomol. Chem., 2014, 12, 8512) (Link)
56. Iron-TEMPO Catalyzed Domino Aerobic Alcohol Oxidation/Oxidative Cross-Dehydrogenative Coupling for the Synthesis of α-Ketoamides
(Surya Srinivas Kotha, S. Chandrasekar, Samrat Sahu and G. Sekar, Eur. J. Org. Chem., 2014, 7415) (Link)
55. Chemoselective reduction of a-keto amides using nickel catalysts (N. Chary Mamillapalli and G. Sekar, Chem. Commun. 2014, 50, 7881) (Link)
54. Isolation and Characterization of Trinuclear Cobalt Complex Containing Trigonal Prismatic Cobalt in Secondary Alcohol Aerobic Oxidation (I. Karthikeyan, S. K. Alamsetti and G. Sekar, Organometallics. 2014, 33, 1665) (Link)
53. Iron(II)Chloride–1,1′-Binaphthyl-2,2′-diamine (FeCl2–BINAM) Complex Catalyzed Domino Synthesis of Bisindolylmethanes from Indoles and Primary Alcohols (S. Badigenchala, D. Ganapathy, A. Das, R. Singh, G. Sekar, Synthesis, 2014, 101)
2013
52. Enantioselective Oxidative Coupling of 2-Naphthol Derivatives by Cu-BINAM-TEMPO Catalyst (G. Sekar, S. K. Alamsetti, E. Poonguzhali and D. Ganapathy, Adv. Synth. Cat. 2013, 355, 2803) (Link)
51. Palladium nanoparticles stabilized by metal-carbon covalent bond: an efficient and reusable nanocatalyst in cross-coupling reactions (D.Ganapathy, and G. Sekar, Cat. Commun. 2013, 39, 50). (Link)
50. Cu-catalyzed in situ generation of thiol using xanthate as a thiol surrogate for the one-pot synthesis of benzothiazoles and benzothiophenes (D. J. C. Prasad and G. Sekar, Org. Biomol. Chem., 2013, 11, 1659). (Link)
2012
49. Domino aziridine ring opening and Buchwald-Hartwig type coupling-cyclization by Palladium Catalyst. (R. Koteshwar Rao, I. Karthikeyan, G.Sekar, Tetrahedron, 2012, 68, 9090)
48. An efficient synthesis of α-hydroxy phosphonates and 2-nitroalkanols using Ba(OH)2 as catalyst (P. Muthupandi, Prem K. Chanani, and G. Sekar, Applied Catalysis A: General, 2012, 441-442, 119)
47. Synthesis of unusual dinuclear chiral iron complex and its application in asymmetric hydrophosphorylation of aldehydes (P. Muthupandi and G. Sekar, Org. Biomol. Chem., 2012, 534). Our artwork related to this article is featured in inside front cover of Organic & Bimolecular Chemistry (Issue 28) (Link)
2011
46. D-Glucosamine as an efficient ligand for copper catalyzed selective synthesis of aniline from aryl halides and NaN3. (K. G. Thakur, K. S. Srinivas, K. Chiranjeevi and G. Sekar, Green Chem. 2011, 13, 2326) (Link)
45. Synthesis of Optically Active 1,4-Benzoxazine Derivatives using Palladium-Catalyzed Coupling Kinetic Resolution (R. Koteshwar Rao and G. Sekar, Tetrahedron: Asymmetry, 2011, 22, 948) (Link)
44. Copper(I)-BINOL Catalyzed Domino Synthesis of 1,4-Benzoxathiines through C(aryl)-O Bond Formation (Invited Paper) (C. Korupalli, A. Dandapat, D. J. C. Prasad and G. Sekar, Organic Chem. Int., 2011, Volume 2011 (2011), Article ID 980765, 7 pages (doi:10.1155/2011/980765) ) (Invited Article) (Link)
43. Zinc-catalyzed aerobic oxidation of benzoins and its extension to enantioselective oxidation (P. Muthupandi and G. Sekar, Tetrahedron Letters, 2011, 52, 692), (Link)
42. An Efficient CuI Catalyzed Synthesis of Diaryl Selenides through C(aryl)-Se Bond Formation using Solvent Acetonitrile as Ligand (Arpan Dandapat, C. Korupalli, D. J. C. Prasad, Rahul Singh and G. Sekar, Synthesis, 2011, 2297)
41. Chiral Zinc-catalyzed aerobic oxidative kinetic resolution of a-hydroxy ketones (P. Muthupandi and G. Sekar, Tetrahedren: Asymmetry, 2011, 22, 522) (Link)
40. D-Glucose as green ligand for selective copper-catalyzed phenol synthesis from aryl halides with an easy catalyst removal. (K. G. Thakur and G. Sekar, Chem. Commun. 2011, 47, 6692). (Link)
39. D-Glucosamine as a green ligand for copper catalyzed synthesis of primary aryl amines from aryl halides and ammonia (K. G. Thakur and G. Sekar, Chem. Commun. 2011, 47, 5076). (Link)
38. Cu-Catalyzed One-Pot Synthesis of Unsymmetrical Diaryl Thioethers by Coupling of Aryl Halides Using Thiol Precursor (D. J. C. Prasad and G. Sekar, Org. Let. 2011, 13, 1008). (This article was highlighted in Organic Chemistry Portal Abstracts), (Link)
2010
37. Chiral cobalt-catalyzed enantioselective aerobic oxidation of -hydroxy esters (S. K. Alamsetti and G. Sekar, Chem. Commun. 2010, 46, 7235. (Link)
36. An efficient copper(II) catalyzed synthesis of benzothiazoles through intramolecular coupling cyclization of N-(2-chlorophenyl)benzothioamides (E. A. Jaseer, D. J. C. Prasad, Arpan Dandapat, G. Sekar, Tetrahedron Letter, 2010, 51, 5009).
35. Cu(I) Catalyzed Intramolecular C(aryl)-O Bond Forming Cyclization for the Synthesis of 1,4-Benzodioxins and its Application in Total Synthesis of Sweetening Isovanilines (Ajay B. Naidu, D. Ganapathy and Govindasamy Sekar, Synthesis, 2010, 3509)
34. Halogenative kinetic resolution of β-aryloxy cyclic alcohols: chiral BINAP mediated SN2 displacement of hydroxy groups by chlorides with inversion of stereochemistry (E. A. Jaseer, I. Karthikeyan and Govindasamy Sekar, Tetrahedron: Asymmetry, 2010, 21, 2177), (Link)
33. Halogenative Kinetic Resolution of -amido Alcohols: Chiral BINAP Mediated SN2 Displacement of Hydroxy Groups by Chlorides with Inversion of Stereochemistry (E. A. Jaseer and G. Sekar, Tetrahedron: Asymmetry, 2010, 21, 780), (Link)
32. Domino Synthesis of 2-Arylbenzo[b]furans by Copper(II)- Catalyzed Coupling of o-Iodophenols and Aryl Acetylenes (E. A. Jaseer, D. J. C. Prasad and G. Sekar, Tetrahedron, 2010, 66, 2077).
31. An Efficient CuCl Catalyzed Selective and Direct Oxidation of β- and γ-Substituted Aliphatic Primary Alcohols to Carboxylic Acids (S. Mannam, and G. Sekar, Synth. Commun. 2010, 40, 2822). (Link)
30. An Efficient Ullmann-type Coupling through C(aryl)-O Bond Forming Intramolecular Cyclization by BINAM Cu(II) Catalyst for the Synthesis of Benzoxazoles (A. B.Naidu, and G. Sekar, Synthesis, 2010, 579). (Link)
29. An Efficient, Mild and Intermolecular Ullmann-Type Synthesis of Thioethers Catalyzed by Diol-Copper(I) Complex (D.J.C. Prasad, and G. Sekar, Synthesis, 2010, 79). (Link)
2009
28. An Efficient Copper Catalyzed Synthesis of Hexahydro-1H-phenothiazines ( D.J.C. Prasad, and G. Sekar, Org. Biomol. Chem., 2009, 5091). (Link)
27. Copper(I) Catalyzed c(aryl)-C(alkynyl) bond formation of aryl iodides with terminal alkynes (K. G. Thakur, and G. Sekar, Synthesis, 2009, 2785). (Link)
26. A general, mild and intermolecular Ullmann-type synthesis of diaryl and alkyl aryl ethers catalyzed by diol-copper(I) complex (A. B. Naidu, E. A. Jaseer and G. Sekar, J. Org. Chem, 2009, 74, 3675) (One of the most accessed top 10 articles in J. org. Chem. for months of April-June, 2009). (Link)
25. An efficient, mild and selective Ullmann-type N-arylation of indoles catalyzed by copper(I) complex (R. K. Rao, A. B. Naidu, E. A. Jaseer and G. Sekar, Tetrahedron, 2009, 65, 4619)
24. An efficient copper(I) complex catalyzed Sonogashira type cross-coupling of aryl halides with terminal alkynes (K. G. Thakur, E. A. Jaseer, A. B. Naidu and G. Sekar, Tetrahedron Lett., 2009, 50, 2965) (One of the most accessed top 25 hot Science Direct articles for months of April- June 2009).
23. Chiral iron complex catalyzed enantioselective oxidation of racemic benzoins (P. Muthupandi, S. K.Alamsetti and G. Sekar, Chem. Commun.. 2009, 3288). (Link)
22. Chiral cobalt catalyzed enantiomer-differentiating oxidation of racemic benzoins using molecular oxygen as stoichiometric oxidant (S. K. Alamsetti, P. Muthupandi and G. Sekar, Chem. Eur. J. 2009, 15, 5424). (Link)
21. Highly efficient copper catalyzed domino ring opening and Goldburg coupling cyclization for the synthesis of 3, 4-dihydro-2H-1,4-benzoxazines (R. K. Rao, A. B. Naidu, and G. Sekar, Org Lett. 2009, 11, 1923) (One of the most accessed top 20 papers in org. Lett. for months of April, 2009)
20. An enantiopure galactose oxidase model: synthesis of chiral amino alcohols through oxidative kinetic resolution catalyzed by chiral copper complex (S. Mannam, and G. Sekar, Tetrahedron: Asymmetry 2009, 20, 495)
19. An efficient intermolecular C(aryl)-S bond forming reaction catalyzed by BINAM-copper(II) complex (D. J. C. Prasad, Ajay B. Naidu and G. Sekar, Tetrahedron Lett., 2009, 50, 1411).
18. Galactose Oxidase Model: Biomimetic, Enantiomer-Differentiating Oxidation of Alcohols by Chiral Copper Complex (S. K. Alamsetti, S. Mannam, P. Muthupandi and G. Sekar, Chem. Eur. J. 2009, 15, 1086) (Link)
2008
17. An efficient intermolecular BINAM-copper (I) catalyzed Ullmann type coupling of aryl iodide with aliphatic alcohols (A. B. Naidu, and G. Sekar, Tetrahedron Lett. 2008, 49, 3147). (Link)
16. CuCl catalyzed selective oxidation of primary alcohols to carboxylic acids with tert-butyl hydroperoxide at room temperature (S. Mannam, and G. Sekar, Tetrahedron Lett. 2008, 49, 2457) (Link)
15. An efficient BINAM-Copper(II) catalyzed Ullmann-type synthesis of diaryl ether (A. B. Naidu, O. R. Ragunath, D. J. C. Prasad and G. Sekar, Tetrahedron Lett. 2008, 49, 1057). (Link)
14. CuCl catalyzed oxidation of aldehydes to carboxylic acids with aqueous tert-butyl hydroperoxide under mild conditions (S. Mannam, and G. Sekar, Tetrahedron Lett. 2008, 49, 1083).
2007
13. Aerobic, Chemoselective Oxidation of Alcohols to Carbonyl Compounds Catalyzed by DABCO-Copper Complex under Mild Conditions (S. Mannam, S. K. Alamsetti and G. Sekar, Adv. Synth. Catal., 2007, 349, 2253). (Link)
12. Highly stereoselective chlorination of -substituted cyclic alcohols using PPh3–NCS: factors that control the stereoselectivity (E. A. Jaseer, A. B. Naidu, S. S. Kumar, R. K. Rao, K. G. Thakur and G. Sekar, Chem. Commun. 2007, 867).
During Ph.D. and Post-Doc
11. Catalyst-Controlled Stereoselective Combinatorial Synthesis (L. F. Tietze, N. Rackelmann and G. Sekar Angew. Chem. Int. Ed. 2002, 42, 4254). (Link)
10. Nonenzymatic kinetic resolution of -amino alcohols: chiral BINAP mediated SN2 displacement of hydroxy groups by halogens through formation of an aziridinium ion intermediate. (G. Sekar and H. Nishiyama Chem. Commun. 2001, 1314). (Link)
9. Nonenzymatic kinetic resolution of secondary alcohols: enantioselective SN2 displacement of hydroxyl groups by halogens in the presence of chiral BINAP. (G. Sekar and H. Nishiyama J. Am. Chem. Soc. 2001, 123, 3603)
8. An efficient method for opening of non-activated aziridines with TMS azide: application in the synthesis of chiral 1, 2-diaminocyclohexane (M. Chandrasekhar, G. Sekar and V. K. Singh Tetrahedron Lett. 2000, 41, 10079)
7. An efficient method for the cleavage of aziridines with hydroxyl compounds. (B. A. Bhanu Prasad, G. Sekar and V. K. Singh Tetrahedron Lett. 2000, 41, 4677)
6. Enantiomerically pure -amino alcohols by enzymatic resolution. (G. Sekar R. M. Kamble and V. K. Singh Tetrahedron: Asymmetry, 1999, 10, 3663).
5. An Efficient method for cleavage of aziridines with aromatic amines. (G. Sekar and V. K. Singh, J. Org. Chem., 1999, 64, 2537). (Link)
4. An efficient method for cleavage of epoxides with aromatic amines. (G. Sekar and V. K. Singh, J. Org. Chem., 1999, 64, 287). (Link)
3. Asymmetric Kharasch reaction: allylic oxidation of olefins using chiral pyridine bis (diphenyoxazoline)- copper complexes and tert-butyl perbenzoate. (G. Sekar, A. Datta Gupta and V. K. Singh, J. Org. Chem., 1998, 63, 2961).
2. Catalytic enantioselective cyclopropanation of olefins using carbenoid chemistry.(V. K. Singh, A. Datta Gupta and G. Sekar, Synthesis, 1997, 137). (Link)
1. Cu(OTf)2 -DBN/DBU complex as an efficient catalyst for allylic oxidation of olefins with tert-butyl perbenzoate. (G. Sekar, A. Datta Gupta and V. K. Singh, Tetrahedron Lett. 1996, 37, 8435).
LIST OF PUBLICATION


![Graphical abstract: Copper-catalyzed domino synthesis of multi-substituted benzo[b]thiophene through radical cyclization using xanthate as a sulfur surrogate](https://pubs.rsc.org/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D0CC08429H&imageInfo.ImageIdentifier.Year=2021)
![Graphical abstract: Copper-catalyzed double C–S bond formation for the synthesis of 2-acyldihydrobenzo[b]thiophenes and 2-acylbenzo[b]thiophenes](https://pubs.rsc.org/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D0CC04647G&imageInfo.ImageIdentifier.Year=2020)